Making up 49.4% of the mass of the Earth’s crust, oxygen is considered a common chemical element. This is also an important curriculum content in the Chemistry curriculum at both middle and high school levels. The following article will help you synthesize important knowledge about the properties, preparation and important applications of this element in life.
What is the definition of oxygen?
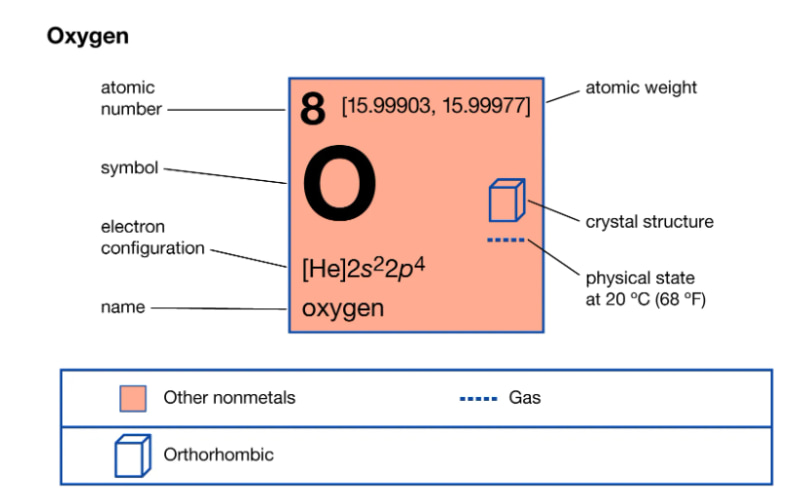
Oxygen (English name is Oxygen) is a chemical element that has symbol is O. The chemical formula of the element (gas) oxygen is O2; The atomic mass is 16 and the molecular mass is 32. Oxygen is known to be the most abundant chemical element in the Earth’s crust. In the form of elemental oxygen compounds found in sugar, water, ores, rocks, human and animal bodies…
Oxygen has atomic number 8, belongs to group VIA, period 2 of the periodic table of chemical elements.
The oxygen atom has an electron configuration of 1s22s22p4, the outermost shell has 6e. Under normal conditions, an oxygen molecule has 2 atoms linked together by a nonpolar covalent bond, the structural formula of oxygen can be written as O=O.
Physical properties of oxygen
To clarify the physical properties of oxygen gas, Chemistry Textbook 8 (Vietnam Education Publishing House) has given observation situations as well as knowledge-provoking questions for you to visualize more clearly.

Observe: There is 1 bottle of oxygen gas with a stopper. Learners’ task is to judge the color of oxygen gas. Then open the stopper of that bottle, bring it close to the nose and use your hand to lightly blow the oxygen into the nose to judge its smell.
The next two questions asked to understand the properties of oxygen are:
1 liter of water at 20 degrees C dissolves 31 ml of oxygen gas. There are gases (eg ammonia) that dissolve 700 liters in 1 liter of water. Is oxygen more or less soluble in water?
Determine whether oxygen is heavier or lighter than air? (The mass ratio of oxygen to air is 32 : 29.)
After answering the above questions and observations, we can easily conclude that the typical physical properties of oxygen gas are:
Oxygen is a gas colorless, odorless, tasteless and heavier than air (d= 32/29, approximately equal to 1.1).
Under atmospheric pressure, the liquid oxidizes at a temperature of -183 degrees Celsius.
Oxygen gas is slightly soluble in water, 100ml of water at 20 degrees Celsius, 1 atm dissolves 3.1ml of oxygen gas.
The solubility of oxygen gas at 20 degrees C and 1 atm is 0.0043g in 100g of H2O.
Chemical properties of oxygen
Learning about oxygen cannot be ignored Chemical properties its basic. In general, oxygen is a very active non-metallic element, especially at high temperatures, easily participating in chemical reactions with many nonmetals, many metals as well as compounds. In chemical compounds the element oxygen has valence II.
Oxygen reacts with metals
Chemistry textbook 8 has used as an example an experiment to prove that oxygen can react with metals as follows:
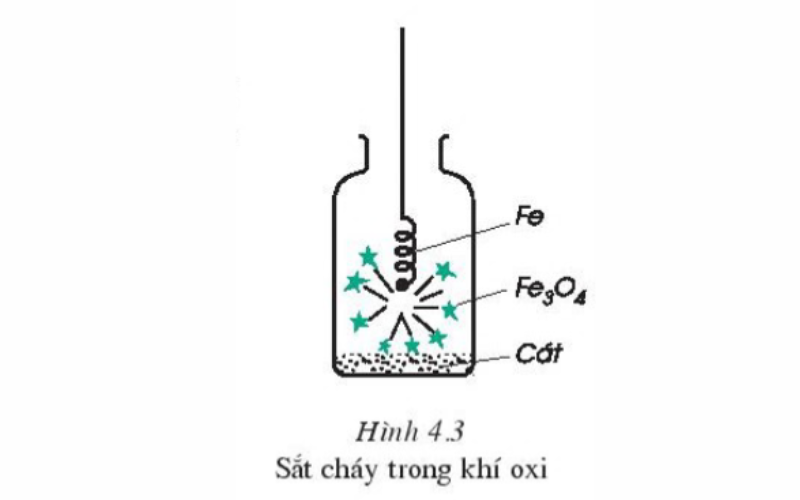
Take a small piece of iron wire and put it into the oxygen cylinder. Observe the experiment to see if any chemical reactions occur. Continue to wrap a piece of wood charcoal on the end of the iron wire, burn the iron and coal to red nóng and then put it in a bottle of oxygen gas. Through observation, we can draw the following observations: When the iron wire wrapped with embers is placed in the oxygen cylinder, the piece of coal burns first, creating a high enough temperature for the iron to burn. Iron burns strongly, brilliantly without flame, without smoke, producing brown molten particles of iron (II, III) oxide, chemical formula is Fe3O4 (ferromagnetic oxide).
The chemical equation goes like this:
3Fe(r) + 2O2(k) → Fe3O4 (Condition: Temperature)
Another example: Mg + O2 → 2MgO (Condition: Temperature)
Oxygen reacts with non-metals
Oxygen reacts with sulfur (S)
Carry out the experiment, put an iron spoon containing a small amount of sulfur powder into the flame of an alcohol lamp to observe. Continue to put the burning sulfur in the bottle containing the oxygen gas and compare the burning of sulfur in oxygen and in air.
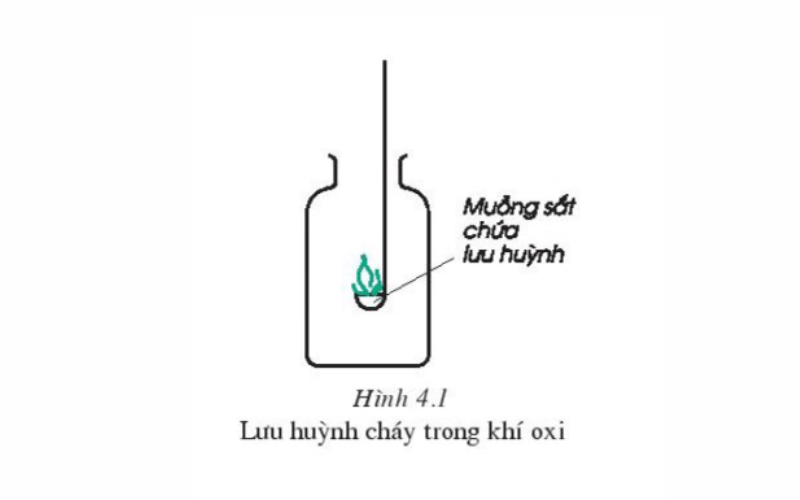
Observing the experiment, we can draw the following observations:
Sulfur burns in the air with a small light blue flame, when burned in the air more intensely to form sulfur dioxide gas SO2 (or sulfur gas) and very little sulfur trioxide (SO3).
The combustion of sulfur in air and oxygen gas is represented by the following chemical equation:
S(r)+ O2(k) → SO2(k) (Condition: Temperature)
Oxygen reacts with phosphorus (P)
Carry out an experiment on adding red phosphorus to an iron spoon (a red-brown solid, insoluble in water). Insert the iron spoon containing phosphorus into the oxygen cylinder. Observe for signs of a chemical reaction. Ignite red phosphorus in air and then quickly put it in a bottle of oxygen and compare the combustion of phosphorus in air and in oxygen. Comment on the ingredients in the vial and the vial wall.
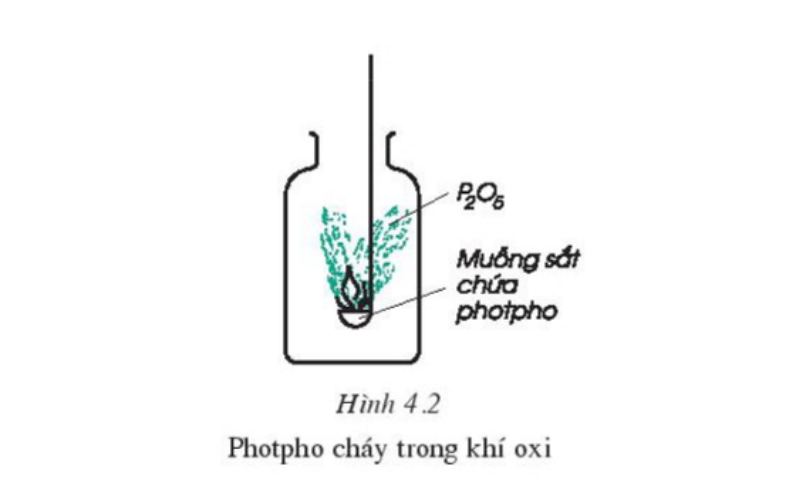
Through the observation process, we can easily draw the following observations: Phosphorus burns strongly in oxygen gas with a bright flame, producing dense white smoke that adheres to the walls of the vial in the form of a water-soluble powder. That white powder is diphosphate pentaoxide, with the chemical formula P2O5.
We have the following chemical equation:
4P(r) + 5O2(k) → 2P2O5(r) (Condition: Temperature)
Carbon burns in the air
C + O2 → CO2 (Condition: Temperature)
Oxygen reacts with compounds
In addition to the action with metals and non-metals, the reaction with compounds is also one of the important chemical properties of oxygen.
Oxygen reacts with methane gas (CH4)
Methane gas (found in pond sludge, biogas) burns in the air by reacting with oxygen gas, giving off a lot of heat. We have PTTH:
CH4(k) + 2O2(k) → CO2(k) + 2H2O(h) (Condition: Temperature)
Oxygen reacts with CO (CO burns in air)
2CO + O2 → 2CO2
Ethanol burns in air
C2H5OH + 3O2 → 2CO2 + 3H2O
See more: What is Iodine? Properties and most popular applications
How to regulate oxygen?
There are many ways to prepare oxygen, depending on its purpose. Here are the most common ways to prepare and produce oxygen in the laboratory and in industry!

Preparation of oxygen in the laboratory
In the laboratory, oxygen gas is prepared by decomposing compounds rich in oxygen and less stable to heat such as KMnO4 (solid), KClO3 (solid)… For example:
2KMnO4 → K2MnO4 + MnO2 + O2 (Condition: Temperature)
Industrial oxygen production
There are two ways to produce oxygen industrially: from air and from water.
Production of oxygen from the air: The air after removing all water, dust, carbon dioxide gas, is liquefied. Fractional distillation of liquid air yields oxygen. Oxygen is transported in steel tanks with a capacity of 100 liters under a pressure of 150 atm.
Production of oxygen from water: Conduct electrolysis of water (water with a small amount of H2SO4 or NaOH dissolved to increase the electrical conductivity of water), we obtain oxygen gas at the anode and hydrogen gas at the cathode. PTTH is as follows:
2H2O → (electrolyte) 2H2 + O2 (k)
Application of oxygen in life
Oxygen gas plays an important role in human life as well as in the fuel burning industry.

The role of oxygen in human and animal life
Oxygen has many important applications decide life of humans and animals. Every day, we need about 20-30m3 of oxygen to sustain life.
Firefighters, pilots or divers need to breathe oxygen stored in special cylinders when on duty.
Application of oxygen in industry – burning fuel
When fuels burn in oxygen, they will generate a much higher temperature than burning in air. Application in the fuel industry of oxygen is also shown in the production of rock-breaking mines, liquid oxygen used to burn fuel in rockets. A fairly close application of oxygen in industry smelting iron and steel is to blow oxygen gas to create a higher temperature, improve the efficiency and quality of cast iron and steel.
Exercises on oxygen in Chemistry Textbook with detailed explanations
After you have mastered the basic theoretical knowledge about oxygen, let’s start practicing to do the exercises to understand the lesson faster. Here are a few oxygen exercises in Chemistry Textbook 8 and 10 Trường THPT Trần Hưng Đạo summarizes with detailed explanations for your reference.

Exercises on oxygen in Chemistry Textbook 8
Lesson 1 (Chemistry Textbook 8, page 84)
Use the appropriate words in the box to fill in the blanks in the following sentences:
Metals, nonmetals, very active, nonmetals very active, compounds.
Oxygen gas is a single element … Oxygen can react with many …, …, …
Suggested answer:
Oxygen gas is a very active non-metallic element. Oxygen can react with many nonmetals, metals, compounds.
Lesson 2 (Chemistry 8, page 84)
Give examples that demonstrate that oxygen is a very active element (especially at high temperatures).
Suggested answer:
Oxygen is a very active element (especially at high temperatures), for example:
3Fe + 2O2 → Fe3O4.
S + O2 → SO2.
Lesson 3 (Chemistry 8, page 84)
Butane with the formula C4H10, when burned, produces carbon dioxide and water vapor, and gives off a lot of heat. Write a chemical equation for the combustion of butane.
Suggested answer:
Chemical equation:
2C4H10 + 13O2 → (temperature) 8CO2 + 10H2O
Lesson 4 (Chemistry Textbook 8, page 84)
Burning 12.4g of phosphorus in a 17g container of oxygen produces diphosphorus pentaoxide P2O5 (a white solid).
a) Which phosphorus or oxygen is left over and how many moles of the substance are left over?
b) What substance is formed? What is the volume?
Suggested answer:
a) We have nP = 12.4/31 = 0.4 (mol); nCO2 = 17/32 = 0.53 (mol).
4P + 5O2 → 2P2O5
Considering the ratio of the number of moles of the problem to the number of moles of the equations of P and O2, we have:
0.4/4 P reacts completely with O2 excess reaction.
nO2 p = 5/4 x nP = 0.5 (mol).
Determine residual nO2 = 0.53 – 0.5 = 0.03 (mol).
b) The substance formed is diphosphate pentaoxide P2O5.
We have nP2O5 = x nP = 0.4/2 = 0.2 mol
mP2O5 = nM = 0.2 x (31x 2 + 16 x 5) = 28.4 (g).
Exercise on oxygen 10 Chemistry Textbook
Exercise 1 (Chemistry 10, page 127)
Match the electron configuration with the appropriate atom.
Electron configuration: A. 1s22s22p5; B. 1s22s22p4; C. 1s22s22p63s23p4; D. 1s22s22p63s23p5
Atoms: a/ Cl; b/ S; c/ O; DF
Suggested answer:
A – d; B – c; C – b; D – a.
Lesson 2 (Chemistry 10, page 127)
Which substance has a nonpolar covalent bond?
A. H2S; B. O2; C. Al2S3; D. SO2.
Suggested answer:
B. O2
Lesson 4 (Chemistry 10, page 127)
Describe the methods of preparing oxygen gas in the laboratory and in industry. Why not apply the method of preparing oxygen in the laboratory for industry and vice versa?
Suggested answer:
Preparation of oxygen in the laboratory:
In the laboratory, oxygen is prepared by decomposing oxygen-rich and low-carbon compounds with heat such as KMnO4, KClO3, …
2KMnO4 → K2MnO4 + MnO2 + O2
2KClO3 → 2KCl + 3O2
Preparation of oxygen in industry:
There are two ways of production:
Production of oxygen from the air: The air after removing all water, dust, carbon dioxide gas, is liquefied. Fractional distillation of liquid air yields oxygen. Oxygen is transported in steel tanks with a capacity of 100 liters under a pressure of 150 atm.
Production of oxygen from water: Conduct electrolysis of water (water with a small amount of H2So2 or NaOH dissolved to increase the conductivity of water), we obtain oxygen gas at the anode and hydrogen gas at the cathode. PTTH is as follows:
2H2O → (electrolyte) 2H2 + O2 (k)
It is not possible to interchange the two processes because industrial preparation requires large amounts of oxygen at a low cost, while in the laboratory, only a small amount is required.
Here is all the information about oxygen Hopefully, it will help you easily synthesize knowledge for effective review. Continue to Trường THPT Trần Hưng Đạo‘s Basic Knowledge column every day to read more interesting articles! Share if you find the article useful, or click “GET UPDATES” at the top of the article to read the earliest weekly shares from Trường THPT Trần Hưng Đạo via email.
[toggle title=”xem thêm thông tin chi tiết về Oxi: Tính chất, ứng dụng và cách điều chế [Tổng hợp Lớp 8 + 10]” state=”close”]
Oxi: Tính chất, ứng dụng và cách điều chế [Tổng hợp Lớp 8 + 10]
Hình Ảnh về: Oxi: Tính chất, ứng dụng và cách điều chế [Tổng hợp Lớp 8 + 10]
Video về: Oxi: Tính chất, ứng dụng và cách điều chế [Tổng hợp Lớp 8 + 10]
Wiki về Oxi: Tính chất, ứng dụng và cách điều chế [Tổng hợp Lớp 8 + 10]
Oxi: Tính chất, ứng dụng và cách điều chế [Tổng hợp Lớp 8 + 10] -
Making up 49.4% of the mass of the Earth's crust, oxygen is considered a common chemical element. This is also an important curriculum content in the Chemistry curriculum at both middle and high school levels. The following article will help you synthesize important knowledge about the properties, preparation and important applications of this element in life.
What is the definition of oxygen?
Oxygen (English name is Oxygen) is a chemical element that has symbol is O. The chemical formula of the element (gas) oxygen is O2; The atomic mass is 16 and the molecular mass is 32. Oxygen is known to be the most abundant chemical element in the Earth's crust. In the form of elemental oxygen compounds found in sugar, water, ores, rocks, human and animal bodies...
Oxygen has atomic number 8, belongs to group VIA, period 2 of the periodic table of chemical elements.
The oxygen atom has an electron configuration of 1s22s22p4, the outermost shell has 6e. Under normal conditions, an oxygen molecule has 2 atoms linked together by a nonpolar covalent bond, the structural formula of oxygen can be written as O=O.
Physical properties of oxygen
To clarify the physical properties of oxygen gas, Chemistry Textbook 8 (Vietnam Education Publishing House) has given observation situations as well as knowledge-provoking questions for you to visualize more clearly.

Observe: There is 1 bottle of oxygen gas with a stopper. Learners' task is to judge the color of oxygen gas. Then open the stopper of that bottle, bring it close to the nose and use your hand to lightly blow the oxygen into the nose to judge its smell.
The next two questions asked to understand the properties of oxygen are:
1 liter of water at 20 degrees C dissolves 31 ml of oxygen gas. There are gases (eg ammonia) that dissolve 700 liters in 1 liter of water. Is oxygen more or less soluble in water?
Determine whether oxygen is heavier or lighter than air? (The mass ratio of oxygen to air is 32 : 29.)
After answering the above questions and observations, we can easily conclude that the typical physical properties of oxygen gas are:
Oxygen is a gas colorless, odorless, tasteless and heavier than air (d= 32/29, approximately equal to 1.1).
Under atmospheric pressure, the liquid oxidizes at a temperature of -183 degrees Celsius.
Oxygen gas is slightly soluble in water, 100ml of water at 20 degrees Celsius, 1 atm dissolves 3.1ml of oxygen gas.
The solubility of oxygen gas at 20 degrees C and 1 atm is 0.0043g in 100g of H2O.
Chemical properties of oxygen
Learning about oxygen cannot be ignored Chemical properties its basic. In general, oxygen is a very active non-metallic element, especially at high temperatures, easily participating in chemical reactions with many nonmetals, many metals as well as compounds. In chemical compounds the element oxygen has valence II.
Oxygen reacts with metals
Chemistry textbook 8 has used as an example an experiment to prove that oxygen can react with metals as follows:

Take a small piece of iron wire and put it into the oxygen cylinder. Observe the experiment to see if any chemical reactions occur. Continue to wrap a piece of wood charcoal on the end of the iron wire, burn the iron and coal to red nóng and then put it in a bottle of oxygen gas. Through observation, we can draw the following observations: When the iron wire wrapped with embers is placed in the oxygen cylinder, the piece of coal burns first, creating a high enough temperature for the iron to burn. Iron burns strongly, brilliantly without flame, without smoke, producing brown molten particles of iron (II, III) oxide, chemical formula is Fe3O4 (ferromagnetic oxide).
The chemical equation goes like this:
3Fe(r) + 2O2(k) → Fe3O4 (Condition: Temperature)
Another example: Mg + O2 → 2MgO (Condition: Temperature)
Oxygen reacts with non-metals
Oxygen reacts with sulfur (S)
Carry out the experiment, put an iron spoon containing a small amount of sulfur powder into the flame of an alcohol lamp to observe. Continue to put the burning sulfur in the bottle containing the oxygen gas and compare the burning of sulfur in oxygen and in air.

Observing the experiment, we can draw the following observations:
Sulfur burns in the air with a small light blue flame, when burned in the air more intensely to form sulfur dioxide gas SO2 (or sulfur gas) and very little sulfur trioxide (SO3).
The combustion of sulfur in air and oxygen gas is represented by the following chemical equation:
S(r)+ O2(k) → SO2(k) (Condition: Temperature)
Oxygen reacts with phosphorus (P)
Carry out an experiment on adding red phosphorus to an iron spoon (a red-brown solid, insoluble in water). Insert the iron spoon containing phosphorus into the oxygen cylinder. Observe for signs of a chemical reaction. Ignite red phosphorus in air and then quickly put it in a bottle of oxygen and compare the combustion of phosphorus in air and in oxygen. Comment on the ingredients in the vial and the vial wall.

Through the observation process, we can easily draw the following observations: Phosphorus burns strongly in oxygen gas with a bright flame, producing dense white smoke that adheres to the walls of the vial in the form of a water-soluble powder. That white powder is diphosphate pentaoxide, with the chemical formula P2O5.
We have the following chemical equation:
4P(r) + 5O2(k) → 2P2O5(r) (Condition: Temperature)
Carbon burns in the air
C + O2 → CO2 (Condition: Temperature)
Oxygen reacts with compounds
In addition to the action with metals and non-metals, the reaction with compounds is also one of the important chemical properties of oxygen.
Oxygen reacts with methane gas (CH4)
Methane gas (found in pond sludge, biogas) burns in the air by reacting with oxygen gas, giving off a lot of heat. We have PTTH:
CH4(k) + 2O2(k) → CO2(k) + 2H2O(h) (Condition: Temperature)
Oxygen reacts with CO (CO burns in air)
2CO + O2 → 2CO2
Ethanol burns in air
C2H5OH + 3O2 → 2CO2 + 3H2O
See more: What is Iodine? Properties and most popular applications
How to regulate oxygen?
There are many ways to prepare oxygen, depending on its purpose. Here are the most common ways to prepare and produce oxygen in the laboratory and in industry!

Preparation of oxygen in the laboratory
In the laboratory, oxygen gas is prepared by decomposing compounds rich in oxygen and less stable to heat such as KMnO4 (solid), KClO3 (solid)... For example:
2KMnO4 → K2MnO4 + MnO2 + O2 (Condition: Temperature)
Industrial oxygen production
There are two ways to produce oxygen industrially: from air and from water.
Production of oxygen from the air: The air after removing all water, dust, carbon dioxide gas, is liquefied. Fractional distillation of liquid air yields oxygen. Oxygen is transported in steel tanks with a capacity of 100 liters under a pressure of 150 atm.
Production of oxygen from water: Conduct electrolysis of water (water with a small amount of H2SO4 or NaOH dissolved to increase the electrical conductivity of water), we obtain oxygen gas at the anode and hydrogen gas at the cathode. PTTH is as follows:
2H2O → (electrolyte) 2H2 + O2 (k)
Application of oxygen in life
Oxygen gas plays an important role in human life as well as in the fuel burning industry.

The role of oxygen in human and animal life
Oxygen has many important applications decide life of humans and animals. Every day, we need about 20-30m3 of oxygen to sustain life.
Firefighters, pilots or divers need to breathe oxygen stored in special cylinders when on duty.
Application of oxygen in industry - burning fuel
When fuels burn in oxygen, they will generate a much higher temperature than burning in air. Application in the fuel industry of oxygen is also shown in the production of rock-breaking mines, liquid oxygen used to burn fuel in rockets. A fairly close application of oxygen in industry smelting iron and steel is to blow oxygen gas to create a higher temperature, improve the efficiency and quality of cast iron and steel.
Exercises on oxygen in Chemistry Textbook with detailed explanations
After you have mastered the basic theoretical knowledge about oxygen, let's start practicing to do the exercises to understand the lesson faster. Here are a few oxygen exercises in Chemistry Textbook 8 and 10 Trường THPT Trần Hưng Đạo summarizes with detailed explanations for your reference.

Exercises on oxygen in Chemistry Textbook 8
Lesson 1 (Chemistry Textbook 8, page 84)
Use the appropriate words in the box to fill in the blanks in the following sentences:
Metals, nonmetals, very active, nonmetals very active, compounds.
Oxygen gas is a single element … Oxygen can react with many …, …, …
Suggested answer:
Oxygen gas is a very active non-metallic element. Oxygen can react with many nonmetals, metals, compounds.
Lesson 2 (Chemistry 8, page 84)
Give examples that demonstrate that oxygen is a very active element (especially at high temperatures).
Suggested answer:
Oxygen is a very active element (especially at high temperatures), for example:
3Fe + 2O2 → Fe3O4.
S + O2 → SO2.
Lesson 3 (Chemistry 8, page 84)
Butane with the formula C4H10, when burned, produces carbon dioxide and water vapor, and gives off a lot of heat. Write a chemical equation for the combustion of butane.
Suggested answer:
Chemical equation:
2C4H10 + 13O2 → (temperature) 8CO2 + 10H2O
Lesson 4 (Chemistry Textbook 8, page 84)
Burning 12.4g of phosphorus in a 17g container of oxygen produces diphosphorus pentaoxide P2O5 (a white solid).
a) Which phosphorus or oxygen is left over and how many moles of the substance are left over?
b) What substance is formed? What is the volume?
Suggested answer:
a) We have nP = 12.4/31 = 0.4 (mol); nCO2 = 17/32 = 0.53 (mol).
4P + 5O2 → 2P2O5
Considering the ratio of the number of moles of the problem to the number of moles of the equations of P and O2, we have:
0.4/4 P reacts completely with O2 excess reaction.
nO2 p = 5/4 x nP = 0.5 (mol).
Determine residual nO2 = 0.53 – 0.5 = 0.03 (mol).
b) The substance formed is diphosphate pentaoxide P2O5.
We have nP2O5 = x nP = 0.4/2 = 0.2 mol
mP2O5 = nM = 0.2 x (31x 2 + 16 x 5) = 28.4 (g).
Exercise on oxygen 10 Chemistry Textbook
Exercise 1 (Chemistry 10, page 127)
Match the electron configuration with the appropriate atom.
Electron configuration: A. 1s22s22p5; B. 1s22s22p4; C. 1s22s22p63s23p4; D. 1s22s22p63s23p5
Atoms: a/ Cl; b/ S; c/ O; DF
Suggested answer:
A - d; B - c; C - b; D - a.
Lesson 2 (Chemistry 10, page 127)
Which substance has a nonpolar covalent bond?
A. H2S; B. O2; C. Al2S3; D. SO2.
Suggested answer:
B. O2
Lesson 4 (Chemistry 10, page 127)
Describe the methods of preparing oxygen gas in the laboratory and in industry. Why not apply the method of preparing oxygen in the laboratory for industry and vice versa?
Suggested answer:
Preparation of oxygen in the laboratory:
In the laboratory, oxygen is prepared by decomposing oxygen-rich and low-carbon compounds with heat such as KMnO4, KClO3, ...
2KMnO4 → K2MnO4 + MnO2 + O2
2KClO3 → 2KCl + 3O2
Preparation of oxygen in industry:
There are two ways of production:
Production of oxygen from the air: The air after removing all water, dust, carbon dioxide gas, is liquefied. Fractional distillation of liquid air yields oxygen. Oxygen is transported in steel tanks with a capacity of 100 liters under a pressure of 150 atm.
Production of oxygen from water: Conduct electrolysis of water (water with a small amount of H2So2 or NaOH dissolved to increase the conductivity of water), we obtain oxygen gas at the anode and hydrogen gas at the cathode. PTTH is as follows:
2H2O → (electrolyte) 2H2 + O2 (k)
It is not possible to interchange the two processes because industrial preparation requires large amounts of oxygen at a low cost, while in the laboratory, only a small amount is required.
Here is all the information about oxygen Hopefully, it will help you easily synthesize knowledge for effective review. Continue to Trường THPT Trần Hưng Đạo's Basic Knowledge column every day to read more interesting articles! Share if you find the article useful, or click “GET UPDATES” at the top of the article to read the earliest weekly shares from Trường THPT Trần Hưng Đạo via email.
[rule_{ruleNumber}]
[box type=”note” align=”” class=”” docs-internal-guid-0462a1df-7fff-150a-ec03-96575bead81f”>
Making up 49.4% of the mass of the Earth’s crust, oxygen is considered a common chemical element. This is also an important curriculum content in the Chemistry curriculum at both middle and high school levels. The following article will help you synthesize important knowledge about the properties, preparation and important applications of this element in life.
What is the definition of oxygen?
Oxygen (English name is Oxygen) is a chemical element that has symbol is O. The chemical formula of the element (gas) oxygen is O2; The atomic mass is 16 and the molecular mass is 32. Oxygen is known to be the most abundant chemical element in the Earth’s crust. In the form of elemental oxygen compounds found in sugar, water, ores, rocks, human and animal bodies…

Oxygen has atomic number 8, belongs to group VIA, period 2 of the periodic table of chemical elements.
The oxygen atom has an electron configuration of 1s22s22p4, the outermost shell has 6e. Under normal conditions, an oxygen molecule has 2 atoms linked together by a nonpolar covalent bond, the structural formula of oxygen can be written as O=O.
Physical properties of oxygen
To clarify the physical properties of oxygen gas, Chemistry Textbook 8 (Vietnam Education Publishing House) has given observation situations as well as knowledge-provoking questions for you to visualize more clearly.

Observe: There is 1 bottle of oxygen gas with a stopper. Learners’ task is to judge the color of oxygen gas. Then open the stopper of that bottle, bring it close to the nose and use your hand to lightly blow the oxygen into the nose to judge its smell.
The next two questions asked to understand the properties of oxygen are:
1 liter of water at 20 degrees C dissolves 31 ml of oxygen gas. There are gases (eg ammonia) that dissolve 700 liters in 1 liter of water. Is oxygen more or less soluble in water?
Determine whether oxygen is heavier or lighter than air? (The mass ratio of oxygen to air is 32 : 29.)
After answering the above questions and observations, we can easily conclude that the typical physical properties of oxygen gas are:
Oxygen is a gas colorless, odorless, tasteless and heavier than air (d= 32/29, approximately equal to 1.1).
Under atmospheric pressure, the liquid oxidizes at a temperature of -183 degrees Celsius.
Oxygen gas is slightly soluble in water, 100ml of water at 20 degrees Celsius, 1 atm dissolves 3.1ml of oxygen gas.
The solubility of oxygen gas at 20 degrees C and 1 atm is 0.0043g in 100g of H2O.
Chemical properties of oxygen
Learning about oxygen cannot be ignored Chemical properties its basic. In general, oxygen is a very active non-metallic element, especially at high temperatures, easily participating in chemical reactions with many nonmetals, many metals as well as compounds. In chemical compounds the element oxygen has valence II.
Oxygen reacts with metals
Chemistry textbook 8 has used as an example an experiment to prove that oxygen can react with metals as follows:

Take a small piece of iron wire and put it into the oxygen cylinder. Observe the experiment to see if any chemical reactions occur. Continue to wrap a piece of wood charcoal on the end of the iron wire, burn the iron and coal to red hot and then put it in a bottle of oxygen gas. Through observation, we can draw the following observations: When the iron wire wrapped with embers is placed in the oxygen cylinder, the piece of coal burns first, creating a high enough temperature for the iron to burn. Iron burns strongly, brilliantly without flame, without smoke, producing brown molten particles of iron (II, III) oxide, chemical formula is Fe3O4 (ferromagnetic oxide).
The chemical equation goes like this:
3Fe(r) + 2O2(k) → Fe3O4 (Condition: Temperature)
Another example: Mg + O2 → 2MgO (Condition: Temperature)
Oxygen reacts with non-metals
Oxygen reacts with sulfur (S)
Carry out the experiment, put an iron spoon containing a small amount of sulfur powder into the flame of an alcohol lamp to observe. Continue to put the burning sulfur in the bottle containing the oxygen gas and compare the burning of sulfur in oxygen and in air.

Observing the experiment, we can draw the following observations:
Sulfur burns in the air with a small light blue flame, when burned in the air more intensely to form sulfur dioxide gas SO2 (or sulfur gas) and very little sulfur trioxide (SO3).
The combustion of sulfur in air and oxygen gas is represented by the following chemical equation:
S(r)+ O2(k) → SO2(k) (Condition: Temperature)
Oxygen reacts with phosphorus (P)
Carry out an experiment on adding red phosphorus to an iron spoon (a red-brown solid, insoluble in water). Insert the iron spoon containing phosphorus into the oxygen cylinder. Observe for signs of a chemical reaction. Ignite red phosphorus in air and then quickly put it in a bottle of oxygen and compare the combustion of phosphorus in air and in oxygen. Comment on the ingredients in the vial and the vial wall.

Through the observation process, we can easily draw the following observations: Phosphorus burns strongly in oxygen gas with a bright flame, producing dense white smoke that adheres to the walls of the vial in the form of a water-soluble powder. That white powder is diphosphate pentaoxide, with the chemical formula P2O5.
We have the following chemical equation:
4P(r) + 5O2(k) → 2P2O5(r) (Condition: Temperature)
Carbon burns in the air
C + O2 → CO2 (Condition: Temperature)
Oxygen reacts with compounds
In addition to the action with metals and non-metals, the reaction with compounds is also one of the important chemical properties of oxygen.
Oxygen reacts with methane gas (CH4)
Methane gas (found in pond sludge, biogas) burns in the air by reacting with oxygen gas, giving off a lot of heat. We have PTTH:
CH4(k) + 2O2(k) → CO2(k) + 2H2O(h) (Condition: Temperature)
Oxygen reacts with CO (CO burns in air)
2CO + O2 → 2CO2
Ethanol burns in air
C2H5OH + 3O2 → 2CO2 + 3H2O
See more: What is Iodine? Properties and most popular applications
How to regulate oxygen?
There are many ways to prepare oxygen, depending on its purpose. Here are the most common ways to prepare and produce oxygen in the laboratory and in industry!

Preparation of oxygen in the laboratory
In the laboratory, oxygen gas is prepared by decomposing compounds rich in oxygen and less stable to heat such as KMnO4 (solid), KClO3 (solid)… For example:
2KMnO4 → K2MnO4 + MnO2 + O2 (Condition: Temperature)
Industrial oxygen production
There are two ways to produce oxygen industrially: from air and from water.
Production of oxygen from the air: The air after removing all water, dust, carbon dioxide gas, is liquefied. Fractional distillation of liquid air yields oxygen. Oxygen is transported in steel tanks with a capacity of 100 liters under a pressure of 150 atm.
Production of oxygen from water: Conduct electrolysis of water (water with a small amount of H2SO4 or NaOH dissolved to increase the electrical conductivity of water), we obtain oxygen gas at the anode and hydrogen gas at the cathode. PTTH is as follows:
2H2O → (electrolyte) 2H2 + O2 (k)
Application of oxygen in life
Oxygen gas plays an important role in human life as well as in the fuel burning industry.

The role of oxygen in human and animal life
Oxygen has many important applications decide life of humans and animals. Every day, we need about 20-30m3 of oxygen to sustain life.
Firefighters, pilots or divers need to breathe oxygen stored in special cylinders when on duty.
Application of oxygen in industry – burning fuel
When fuels burn in oxygen, they will generate a much higher temperature than burning in air. Application in the fuel industry of oxygen is also shown in the production of rock-breaking mines, liquid oxygen used to burn fuel in rockets. A fairly close application of oxygen in industry smelting iron and steel is to blow oxygen gas to create a higher temperature, improve the efficiency and quality of cast iron and steel.
Exercises on oxygen in Chemistry Textbook with detailed explanations
After you have mastered the basic theoretical knowledge about oxygen, let’s start practicing to do the exercises to understand the lesson faster. Here are a few oxygen exercises in Chemistry Textbook 8 and 10 Trường THPT Trần Hưng Đạo summarizes with detailed explanations for your reference.

Exercises on oxygen in Chemistry Textbook 8
Lesson 1 (Chemistry Textbook 8, page 84)
Use the appropriate words in the box to fill in the blanks in the following sentences:
Metals, nonmetals, very active, nonmetals very active, compounds.
Oxygen gas is a single element … Oxygen can react with many …, …, …
Suggested answer:
Oxygen gas is a very active non-metallic element. Oxygen can react with many nonmetals, metals, compounds.
Lesson 2 (Chemistry 8, page 84)
Give examples that demonstrate that oxygen is a very active element (especially at high temperatures).
Suggested answer:
Oxygen is a very active element (especially at high temperatures), for example:
3Fe + 2O2 → Fe3O4.
S + O2 → SO2.
Lesson 3 (Chemistry 8, page 84)
Butane with the formula C4H10, when burned, produces carbon dioxide and water vapor, and gives off a lot of heat. Write a chemical equation for the combustion of butane.
Suggested answer:
Chemical equation:
2C4H10 + 13O2 → (temperature) 8CO2 + 10H2O
Lesson 4 (Chemistry Textbook 8, page 84)
Burning 12.4g of phosphorus in a 17g container of oxygen produces diphosphorus pentaoxide P2O5 (a white solid).
a) Which phosphorus or oxygen is left over and how many moles of the substance are left over?
b) What substance is formed? What is the volume?
Suggested answer:
a) We have nP = 12.4/31 = 0.4 (mol); nCO2 = 17/32 = 0.53 (mol).
4P + 5O2 → 2P2O5
Considering the ratio of the number of moles of the problem to the number of moles of the equations of P and O2, we have:
0.4/4 P reacts completely with O2 excess reaction.
nO2 p = 5/4 x nP = 0.5 (mol).
Determine residual nO2 = 0.53 – 0.5 = 0.03 (mol).
b) The substance formed is diphosphate pentaoxide P2O5.
We have nP2O5 = x nP = 0.4/2 = 0.2 mol
mP2O5 = nM = 0.2 x (31x 2 + 16 x 5) = 28.4 (g).
Exercise on oxygen 10 Chemistry Textbook
Exercise 1 (Chemistry 10, page 127)
Match the electron configuration with the appropriate atom.
Electron configuration: A. 1s22s22p5; B. 1s22s22p4; C. 1s22s22p63s23p4; D. 1s22s22p63s23p5
Atoms: a/ Cl; b/ S; c/ O; DF
Suggested answer:
A – d; B – c; C – b; D – a.
Lesson 2 (Chemistry 10, page 127)
Which substance has a nonpolar covalent bond?
A. H2S; B. O2; C. Al2S3; D. SO2.
Suggested answer:
B. O2
Lesson 4 (Chemistry 10, page 127)
Describe the methods of preparing oxygen gas in the laboratory and in industry. Why not apply the method of preparing oxygen in the laboratory for industry and vice versa?
Suggested answer:
Preparation of oxygen in the laboratory:
In the laboratory, oxygen is prepared by decomposing oxygen-rich and low-carbon compounds with heat such as KMnO4, KClO3, …
2KMnO4 → K2MnO4 + MnO2 + O2
2KClO3 → 2KCl + 3O2
Preparation of oxygen in industry:
There are two ways of production:
Production of oxygen from the air: The air after removing all water, dust, carbon dioxide gas, is liquefied. Fractional distillation of liquid air yields oxygen. Oxygen is transported in steel tanks with a capacity of 100 liters under a pressure of 150 atm.
Production of oxygen from water: Conduct electrolysis of water (water with a small amount of H2So2 or NaOH dissolved to increase the conductivity of water), we obtain oxygen gas at the anode and hydrogen gas at the cathode. PTTH is as follows:
2H2O → (electrolyte) 2H2 + O2 (k)
It is not possible to interchange the two processes because industrial preparation requires large amounts of oxygen at a low cost, while in the laboratory, only a small amount is required.
Here is all the information about oxygen Hopefully, it will help you easily synthesize knowledge for effective review. Continue to Trường THPT Trần Hưng Đạo‘s Basic Knowledge column every day to read more interesting articles! Share if you find the article useful, or click “GET UPDATES” at the top of the article to read the earliest weekly shares from Trường THPT Trần Hưng Đạo via email.
[/box]
#Oxi #Tính #chất #ứng #dụng #và #cách #điều #chế #Tổng #hợp #Lớp
[/toggle]
Bạn thấy bài viết Oxi: Tính chất, ứng dụng và cách điều chế [Tổng hợp Lớp 8 + 10] có khắc phục đươc vấn đề bạn tìm hiểu ko?, nếu ko hãy comment góp ý thêm về Oxi: Tính chất, ứng dụng và cách điều chế [Tổng hợp Lớp 8 + 10] bên dưới để thpttranhungdao.edu.vn có thể thay đổi & cải thiện nội dung tốt hơn cho độc giả nhé! Cám ơn bạn đã ghé thăm Website Trường THPT Trần Hưng Đạo
Phân mục: Giáo dục
#Oxi #Tính #chất #ứng #dụng #và #cách #điều #chế #Tổng #hợp #Lớp


Trả lời